Elidah Secures FDA Clearance for At-Home Device Treating Post-Prostate Surgery Incontinence
BloElidah has received FDA clearance for Elitone for Men, an at-home therapy targeting urinary incontinence after prostate surgery. The device expands non-invasive treatment options and enables earlier intervention outside clinical settings.
REGULATORY
Editor
4/2/20261 min read
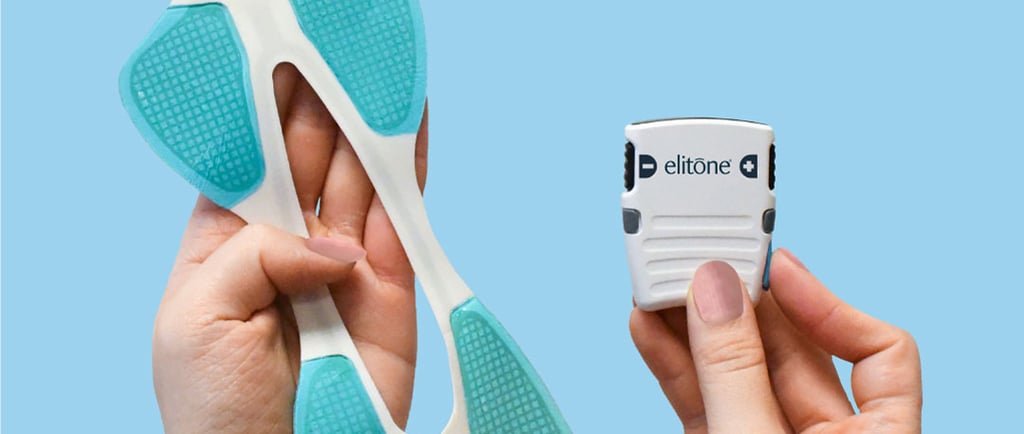

Elidah has announced FDA clearance for Elitone for Men, a non-invasive, at-home treatment designed to address urinary incontinence following prostate surgery. The device is positioned as an over-the-counter therapy and is eligible for Medicare coverage, with US availability expected in April 2026.
Elitone for Men delivers neuromuscular stimulation to strengthen pelvic floor muscles, aiming to improve bladder control. The therapy is designed for daily 20-minute use at home, offering an alternative to clinic-based or invasive interventions. Urinary incontinence remains a common complication after prostate procedures, affecting a large proportion of patients in both the short and longer term.
Clinical data cited by the company shows improved recovery timelines for users of the device. Patients using Elitone reached comparable continence levels in 8 weeks that control groups achieved in 12 weeks. After 12 weeks, users reported a 92% reduction in leakage, compared to 68% in the control group.
The product is part of Elidah’s broader Elitone platform, which focuses on non-invasive pelvic health therapies for both men and women. The company states that the platform has been used by tens of thousands of patients and is also applied to conditions such as overactive bladder.