Bright Uro Receives FDA Clearance for Abdominal Sensor in Bladder Dysfunction Testing
Bright Uro has received FDA clearance for its Glean abdominal sensor, expanding its catheter-free urodynamics platform for evaluating bladder dysfunction. The approval adds multi-channel pressure monitoring capabilities to the company’s Glean system, which was cleared by the FDA in 2025.
REGULATORY
Editor
5/11/20261 min read
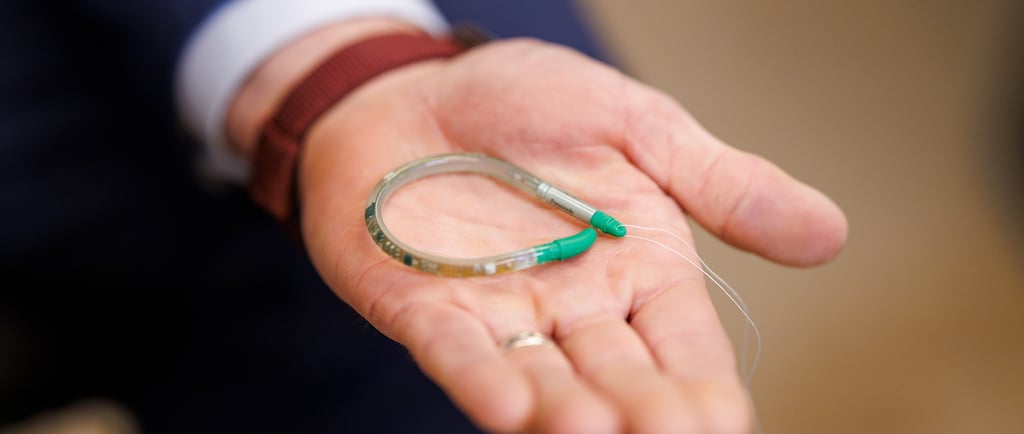

Bright Uro has secured FDA clearance for the abdominal sensor used in its Glean urodynamics system, enabling multi-channel urodynamic and pressure studies for bladder dysfunction evaluation. The approval expands the functionality of the company’s Glean platform, which previously received FDA clearance in March 2025.
The Glean system is designed to assess how the bladder and lower urinary tract store and release urine by measuring pressure and flow characteristics. With the addition of the abdominal sensor, clinicians can now use a catheter-free ambulatory urodynamics protocol to support diagnosis and treatment decisions for patients with lower urinary tract dysfunction and related urological conditions.
The California-based company said the expanded system responds to customer demand for more comprehensive urodynamic testing capabilities. CEO Derek Herrera said the launch reflects the company’s focus on product innovation and meeting clinician needs.
The clearance comes as bladder dysfunction and urinary incontinence continue to attract investment and acquisition activity across medtech. In January 2026, Boston Scientific acquired Valencia Technologies, adding the eCoin tibial nerve stimulator for urge urinary incontinence to its portfolio. The company also completed its $3.3 billion acquisition of Axonics in 2024 to expand its sacral neuromodulation offerings for bladder dysfunction treatment.
According to the National Association for Continence, around 80 million people in the US experience voiding disorders including overactive bladder, urinary incontinence, and benign prostatic hyperplasia.