Bairitone Health Receives FDA Breakthrough Device Designation for Sleep Apnea Platform Somnar
Bairitone Health has received FDA Breakthrough Device Designation for Somnar, a wearable sleep diagnostics platform designed to map airway obstruction during sleep. The designation could accelerate development of the company’s AI-driven approach to obstructive sleep apnea diagnosis and treatment planning.
REGULATORY
Editor
5/8/20261 min read
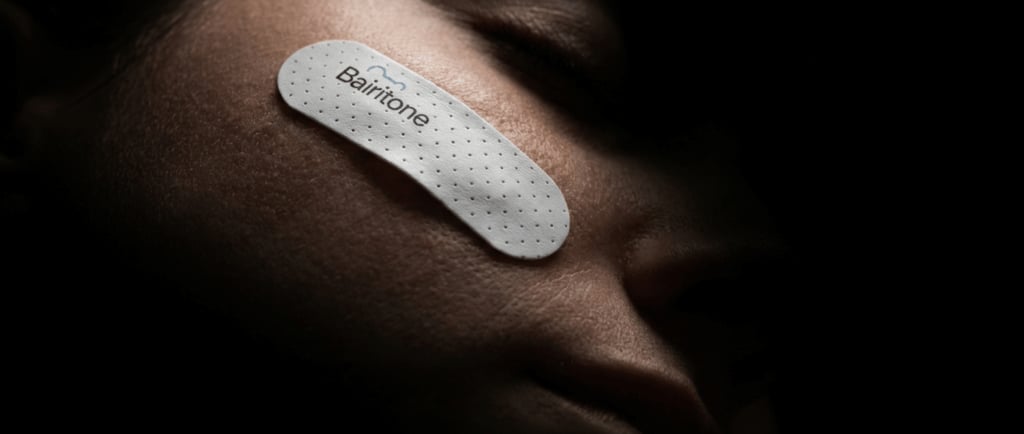
Bairitone Health has received FDA Breakthrough Device Designation for Somnar, its non-invasive sleep diagnostics platform designed to identify the root causes of airway obstruction during sleep.
Somnar combines a skin-mounted facial sensor patch with an AI-powered analysis platform that tracks upper airway behavior overnight. The company describes the system as a “sleep sonar” that captures natural tissue vibrations produced during breathing and converts them into a dynamic map of airway anatomy and function.
Unlike traditional imaging approaches such as MRI, X-ray, or ultrasound, the platform continuously monitors airway behavior across different sleep stages and body positions. The goal is to help clinicians identify where and why airway collapse occurs, supporting more personalized treatment decisions for obstructive sleep apnea and related sleep disorders.
Bairitone Health says the platform represents the first “anatomic polysomnogram.” The system remains in the research and development stage and has not yet been FDA-cleared for commercial sale or clinical use. It is currently being used in investigational and clinical research settings.
The FDA Breakthrough Devices Program is intended to accelerate the development and review of technologies that may provide more effective diagnosis or treatment for serious conditions. The designation gives companies closer interaction with the FDA during the regulatory process while maintaining standard safety and effectiveness requirements.
The milestone follows additional industry recognition for the company. In November 2025, Somnar won the diagnostics category at the inaugural American Academy of Sleep Medicine Sleep Disruptors Innovation Awards.